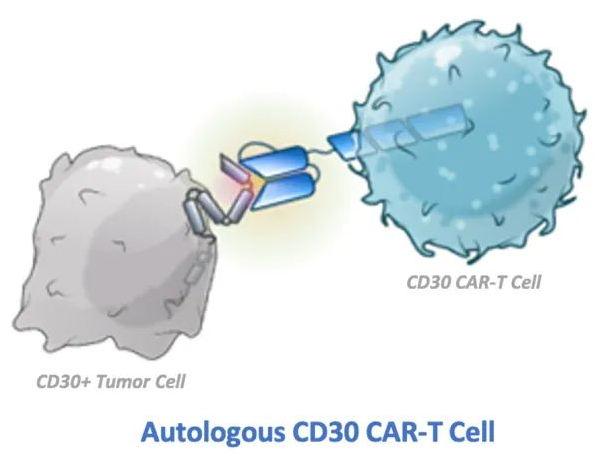
**Patient Story: CAR-T Therapy Brings New Hope for Lymphoma Patients – A Case from a Chinese Hospital** In the field of cancer treatment, China has achieved groundbreaking success with CAR-T cell therapy, especially in treating relapsed or refractory lymphoma. Today, we share the story of one patient whose life was transformed through this revolutionary treatment Read More